Successful PEI Evaluation of Accu-Tell® SARS-CoV-2 Ag Rapid Test for nasopharyngeal sample
Good News: Our Accu-Tell® SARS-CoV-2 Ag Rapid Test for nasopharyngeal sample has successfully passed PEI evaluation by BfARM in Germany. Please check the attached screen shot: ja means yes for successful evaluation.
In our clinical study, ACCU-TELL® SARS-CoV-2 Ag Cassette (Nasopharyngeal Swab) has been evaluated with specimens obtained from the patients. RT-PCR is used as the reference method. Specimens were considered positive if PCR indicated a positive result. The performance characteristics are as following:
Relative Sensitivity: 95.7% (95%CI*:90.3%-98.6%)*
Relative Specificity: 99.2% (95%CI*:97.6%-99.8%)*
Relative accuracy: 98.3% (95%CI*:96.7%-99.3%)*
* Confidence Intervals
The Estimated LOD is 1000 TCID50/mL.
Till now, our following rapid tests relevant to COVID-19 infection diagnosis have got CE registration in EU:
- Accu-Tell® COVID-19 IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma);
- Accu-Tell® SARS-CoV-2 Ag Rapid Test Cassette (Nasopharyngeal Swab);
- Accu-Tell® SARS-CoV-2 + Flu A&B Antigen Combo Cassette (Nasopharyngeal Swab);
- Accu-Tell® SARS-CoV-2 Neutralizing Antibody Cassette (Whole Blood/Serum/Plasma);
- Accu-Tell® COVID-19 Antigen Cassette (Saliva);
- Accu-Tell® COVID-19 Antigen Cassette (Nasal Swab);
- Accu-Tell® SARS-CoV-2 Neutralizing Antibody Cassette (RBD) (Whole Blood/Serum/Plasma)
If you need any of the above COVID-19 Testing products, please feel free to contact us. We will provide the samples for your evaluation.
الصفحة السابقة: What is SARS-CoV-2 Neutralizing Antibody
-
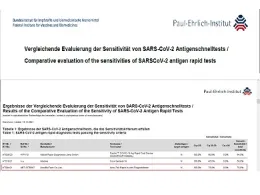
- PEI evaluation result
Jan. 11, 2022
-
- Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern
Dec. 03, 2021




